Description
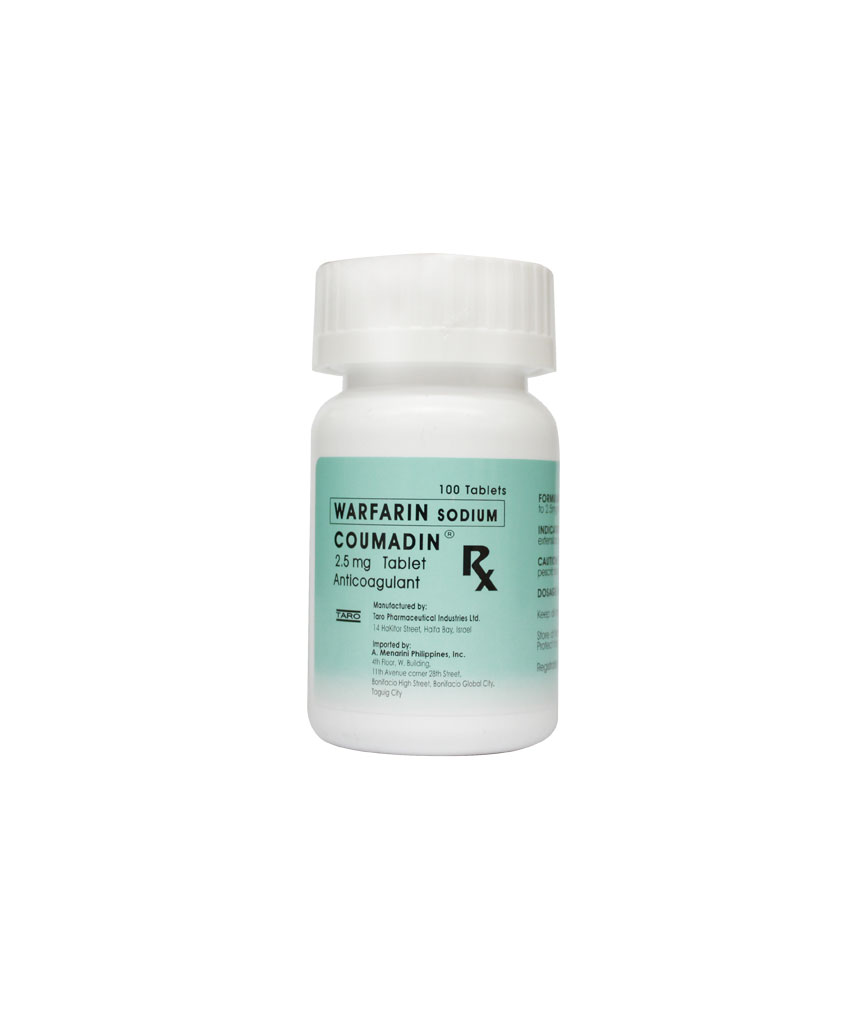
Buy COUMADIN SODIUM 2.5MG TABLET at Rose Pharmacy Online Drugstore
Description 😕Coumadin tablets also contain lactose, starch, magnesium stearate and other ingredients: 1-mg tablet: D&C red 6; 2.5-mg tablet: FD&C Blue 1 and D&C yellow 10; 5-mg tablet: FD&C yellow 6.
Coumadin (crystalline warfarin sodium), a vitamin K-dependent anticoagulant factor, is crystalline sodium warfarin isopropanol clathrate. The crystallization of warfarin sodium virtually eliminates trace impurities present in amorphous warfarin sodium, thus achieving a crystalline product of the highest purity. Warfarin is the coined generic name for 3-(a-acetonyl-benzyl)-4-hydroxycoumarin. Its empirical formula is C19H15NaO4.
Crystalline warfarin sodium occurs as a white, odorless, crystalline powder, discolored by light and very soluble in water; freely soluble in alcohol; very slightly soluble in chloroform and in ether.
Indications / Uses :Prophylaxis and/or treatment of venous thrombosis and its extension, pulmonary embolism, thromboembolic complications associated with atrial fibrillation, cardiac valve replacement and as an adjunct in the prophylaxis of systemic embolism after myocardial infarction.
Administration :May be taken with or without food.
Contraindications :Anticoagulation is contraindicated in any localized or general physical condition or personal circumstance in which the hazard of hemorrhage might be greater than its potential clinical benefits eg, pregnancy. Pregnancy: Women who are or may become pregnant because the drug passes through the placental barrier and may cause fatal hemorrhage to the fetus in utero. Furthermore, there have been reports of birth malformations in children born to mothers who have been treated with warfarin during pregnancy.
Embryopathy characterized by nasal hypoplasia with or without stippled epiphyses (chondrodysplasia punctata) has been reported in pregnant women exposed to warfarin during the 1st trimester. Central nervous system abnormalities also have been reported, including dorsal midline dysplasia characterized by agenesis of the corpus callosum, Dandy-Walker malformation, and midline cerebellar atrophy. Ventral midline dysplasia, characterized by optic atrophy and eye abnormalities has been observed. Mental retardation, blindness, and other central nervous system abnormalities have been reported in association with 2nd and 3rd trimester exposure. Although rare, teratogenic reports following in utero exposure to warfarin include urinary tract anomalies eg, single kidney, asplenia, anencephaly, spina bifida, cranial nerve palsy, hydrocephalus, cardiac defects and congenital heart disease, polydactyly, deformities of toes, diaphragmatic hernia, and corneal leukoma.
Spontaneous abortion and stillbirth are known to occur and a higher risk of fetal mortality is associated with the use of warfarin.
Women of childbearing potential who are candidates for anticoagulant therapy should be carefully evaluated and the indications critically reviewed with the patient. If the patient becomes pregnant while taking this drug, she should be apprised of the potential risks to the fetus, and the possibility of termination of the pregnancy should be discussed in light of those risks.
Hemorrhagic tendencies or blood dyscrasias.
Recent or contemplated surgery of the central nervous system, eye, or traumatic surgery resulting in large open surfaces.
Bleeding tendencies associated with active ulceration or overt bleeding of the gastrointestinal, genitourinary or respiratory tracts; cerebrovascular hemorrhage; aneurysms-cerebral, dissecting aorta; pericarditis, pericardial effusions and bacterial endocarditis.
Threatened abortion, eclampsia and preeclampsia.
Inadequate laboratory facilities or unsupervised senility, alcoholism, psychosis or lack of patient cooperation.
Spinal puncture and other diagnostic or therapeutic procedures with potential for uncontrollable bleeding.
Miscellaneous: Major regional, lumbar block anesthesia and malignant hypertension.
Special Precautions : Periodic determination of PT or other suitable coagulation test is essential.
Numerous factors, alone or in combination, including travel, changes in diet, environment, physical state and medication may influence response of the patient to anticoagulants. It is generally good practice to monitor the patient’s response with additional PT determinations in the period immediately after discharge from the hospital, and whenever other medications are initiated, discontinued or taken haphazardly. The following factors are listed for reference; however, other factors may also affect the anticoagulant response.
The following factors, alone or in combination, may be responsible for increased PT response: Endogenous Factors: Cancer; collagen disease; congestive heart failure; diarrhea; elevated temperature; hepatic disorders (infectious hepatitis, jaundice); hyperthyroidism; poor nutritional state; steatorrhea and vitamin K deficiency.
Exogenous Factors: Acetaminophen, alcohol*; allopurinol; aminosalicylic acid; amiodarone HCl; anabolic steroids; anesthetics (inhalation), antibiotics; bromelains; chenodiol, chloral hydrate*; chlorpropamide; chymotrypsin; cimetidine; clofibrate; Coumadin overdosage; dextran; dextrothyroxine; diazoxide; diflunisal; diuretics*; disulfiram; ethacrynic acid; fenoprofen; fluoroquinolone antibiotics, glucagon; hepatotoxic drugs; ibuprofen, indomethacin; influenza virus vaccine; lovastatin, mefenamic acid; methyldopa; methylphenidate; metronidazole, miconazole; MAOIs; moricizine HCl*; nalidixic acid; naproxen; pentoxifylline; phenylbutazone; phenytoin; prolonged narcotics; propafenone, pyrazolones, quinidine, quinine; ranitidine*; salicylates; sulfinpyrazone; long-acting sulfonamides; sulindac; tamoxifen, thyroid drugs; tolbutamide; trimethoprim/sulfamethoxazole; also, other medications affecting blood elements which may modify hemostasis; dietary deficiencies; prolonged hot weather, unreliable PT determinations.
*Increased and decreased PT responses have been reported.
The following factors, alone or in combination, may be responsible for decreased PT response: Endogenous Factors: Edema; hereditary coumarin resistance; hyperlipemia; hypothyroidism.
Exogenous Factors: Adrenocortical steroids; alcohol*; aminoglutethimide, antacids; antihistamines; barbiturates; carbamazepine; chloral hydrate*; chlordiazepoxide; cholestyramine; Coumadin underdosage; diuretics*; ethchlorvynol; glutethimide; griseofulvin; haloperidol; meprobamate; moricizine HCl*, nafcillin, oral contraceptives; paraldehyde; primidone; ranitidine*; rifampin; sucralfate, trazodone, vitamin C; also, diet high in vitamin K; unreliable PT determinations.
*Increased and decreased PT responses have been reported.
Because a patient may be exposed to a combination of the previously mentioned factors, the net effect of Coumadin on PT response may be unpredictable. More frequent PT monitoring is therefore advisable.
Medications of unknown interaction with coumarins are best regarded with caution when these medications are started or stopped, more frequent PT monitoring is advisable.
Coumarins may also affect the action of other drugs. Hypoglycemic agents (chlorpropamide and tolbutamide) and anticonvulsants (phenytoin and phenobarbital) may accumulate in the body as a result of interference with either their metabolism or excretion.
It has been reported that concomitant administration of warfarin and ticlopidine may be associated with cholestatic hepatitis.
Special Risk Patients: Caution should be observed when warfarin sodium is administered to certain patients eg, the elderly or debilitated or when administered in any situation or physical condition where added risk of hemorrhage is present.
Intramuscular (IM) injections of concomitant medications should be confined to the upper extremities which permits easy access for manual compression, inspections for bleeding and use of pressure bandages.
Caution should be observed when Coumadin (or warfarin) is administered concomitantly with nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, to be certain that no change in anticoagulation dosage is required. In addition to specific drug interactions that might affect PT, NSAIDs, including aspirin, can inhibit platelet aggregation and can cause gastrointestinal bleeding, peptic ulceration and/or perforation.
Information for Patients: The objective of anticoagulant therapy is to control the coagulation mechanism so that thrombosis is prevented while avoiding spontaneous bleeding. Effective therapeutic levels with minimal complications are in part dependent upon cooperative and well-instructed patients who communicate effectively with their physician. Various Coumadin patient educational guides are available to physicians on request.
Patients should be advised: Strict adherence to prescribed dosage schedule is necessary. Do not take or discontinue any other medication, except on advice of physician. Avoid alcohol, salicylates (eg, aspirin and topical analgesics), large amounts of green leafy vegetables and/or drastic changes in dietary habits, which may affect Coumadin therapy. Coumadin may cause a red-orange discoloration of alkaline urine. The patient should notify the physician if any illness eg, diarrhea, infection or fever develops or if any unusual symptoms eg, pain, swelling or discomfort appear or if prolonged bleeding from cuts, increased menstrual flow or vaginal bleeding, nosebleeds or bleeding of gums from brushing, unusual bleeding or bruising, red or dark brown urine, red or tar black stools or diarrhea occurs.











Reviews
There are no reviews yet.