Description
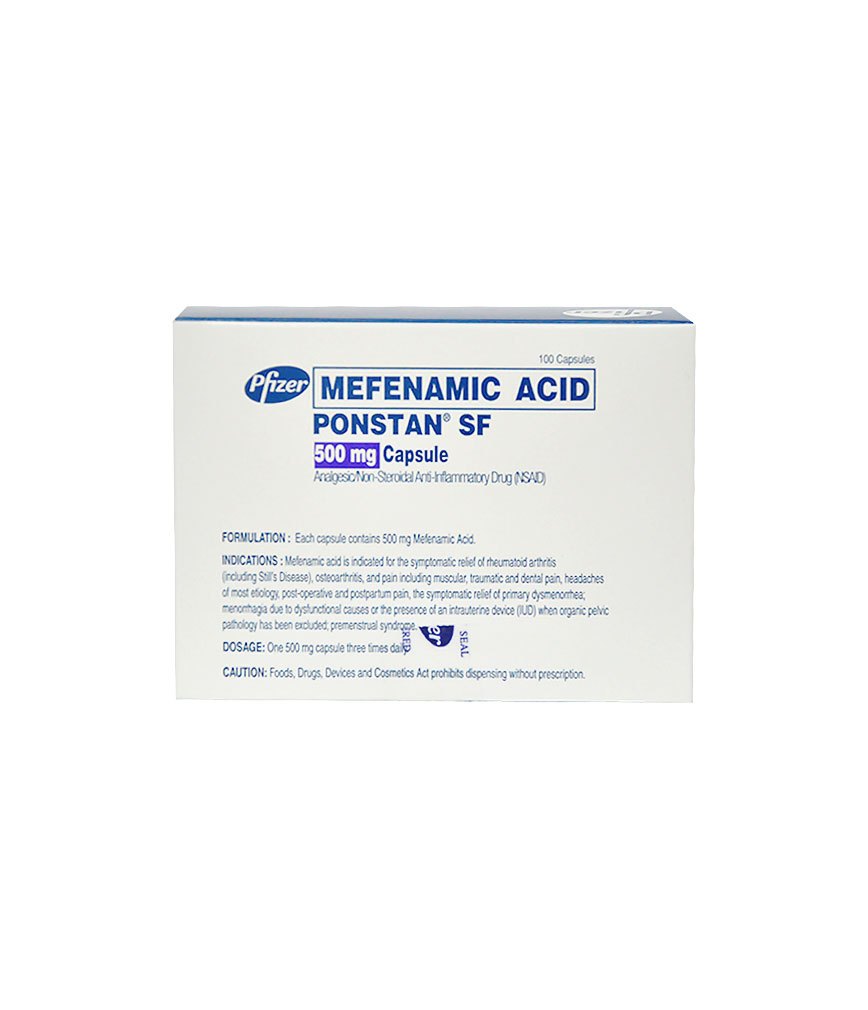
PONSTAN SF 500MG CAPSULE
Description :
Mefenamic acid is N-(2,3,-xylyl)-anthranilic acid. It is an orally active analgesic agent.
It is a white powder with a melting point of 230-231?C, molecular weight of 241.28 and water solubility of 0.004% at pH 7.1.
Indications / Uses :
Symptomatic relief of rheumatoid arthritis (including Still’s disease), osteoarthritis and pain including muscular, traumatic and dental pain, headaches of most etiology and postoperative and postpartum pain.
Symptomatic relief of primary dysmenorrhea.
Menorrhagia due to dysfunctional causes or the presence of an intrauterine device (IUD) when organic pelvic pathology has been excluded.
Premenstrual syndrome.
Relief of pyrexia in pediatric patients >6 months.
Administration :
Should be taken with food: Take immediately after meals.
Contraindications :
Hypersensitivity to mefenamic acid or to any of the excipients of Ponstan eg, lactose, gelatin, sodium lauryl sulfate, purified water, corn starch, corn starch pregelatinized, methyl cellulose, microcrystalline cellulose, silicon dioxide, magnesium stearate, polyethylene glycol, polysorbate 80, saccharin sodium, vanillin, opaspray yellow, wax candelilla FCC powder, veegum, povidone, sodium hydroxide, glucono d lactone, hydrochloric acid, sorbitol solution, sodium benzoate, sodium carboxymethylcellulose granulated sugar, artificial banana flavor, anise mint flavor, alcohol and chocolate flavor.
Because the potential exists for cross-sensitivity to aspirin or other nonsteroidal anti-inflammatory drugs (NSAIDs), Ponstan should not be given to patients in whom these drugs induce symptoms of bronchospasm, allergic rhinitis or urticaria.
Patients with active ulceration or chronic inflammation of either the upper or lower gastrointestinal tract and should be avoided in patients with preexisting renal disease.
Treatment of perioperative pain in the setting of coronary artery bypass graft (CABG) surgery.
Patients with severe renal and hepatic failure.
Patients with severe heart failure.
Special Precautions:
The use of mefenamic acid (Ponstan) with concomitant NSAIDs including COX-2 inhibitors should be avoided. Concomitant use of a systemic NSAID and another systemic NSAID may increase frequency of gastrointestinal ulcers and bleeding.
Cardiovascular Effects: NSAIDs may cause an increased risk of serious cardiovascular thrombotic events, myocardial infarction and stroke which can be fatal. This risk may increase with duration of use. Patients with known cardiovascular disease may be at greater risk. To minimize the potential risk for an adverse cardiovascular event in patients treated with mefenamic acid (Ponstan), the lowest effective dose should be used for the shortest duration possible. Physicians and patients should remain alert for the development of such events, even in the absence of previous cardiovascular symptoms. Patients should be informed about the signs and/or symptoms of serious cardiovascular toxicity and the steps to take if they occur (see Contraindications).
Hypertension: As with all NSAIDS, mefenamic acid (Ponstan) can lead to the onset of new hypertension or worsening of preexisting hypertension, either of which may contribute to the increased incidence of cardiovascular events. NSAIDs, including mefenamic acid (Ponstan), should be used with caution in patients with hypertension. Blood pressure should be monitored closely during the initiation of therapy with mefenamic acid (Ponstan) and throughout the course of therapy.
Fluid Retention and Edema: As with other drugs known to inhibit prostaglandin synthesis, fluid retention and edema have been observed in some patients taking NSAIDs, including mefenamic acid (Ponstan). Therefore, mefenamic acid (Ponstan) should be used with caution in patients with compromised cardiac function and other conditions predisposing to, or worsened by, fluid retention. Patients with preexisting congestive heart failure or hypertension should be closely monitored.
Gastrointestinal (GI) Effects: If diarrhea occurs, the dosage should be reduced or temporarily suspended. Symptoms may recur in certain patients following subsequent exposure.
NSAIDs including mefenamic acid (Ponstan), can cause serious GI adverse events including inflammation, bleeding, ulceration and perforation of the stomach, small intestine or large intestine, which can be fatal. When GI bleeding or ulceration occurs in patients receiving mefenamic acid (Ponstan), the treatment should be withdrawn. Patients most at risk of developing these types of GI complications with NSAIDs are the elderly, patients with cardiovascular disease, patients using concomitant aspirin, or patients with a prior history of, or active GI disease eg, ulceration, GI bleeding or inflammatory conditions. Therefore, mefenamic acid (Ponstan) should be used with caution in these patients (see Contraindications).
Skin Reactions: Serious skin reactions, some of them fatal including exfoliative dermatitis, Stevens-Johnson syndrome and toxic epidermal necrolysis, have been reported very rarely in association with the use of NSAIDs including mefenamic acid (Ponstan). Patients appear to be at highest risk for these events early in the course of therapy, the onset of the event occurring in the majority of cases within the 1st month of treatment. Mefenamic acid (Ponstan) should be discontinued at the 1st appearance of skin rash, mucosal lesions or any other sign of hypersensitivity.
Laboratory Tests: A false-positive reaction for urinary bile, using the diazo tablet test, may result following mefenamic acid (Ponstan) administration. If biliuria is suspected, other diagnostic procedures eg, the Harrison spot test, should be performed.
Renal Effects: In rare cases, NSAIDs, including mefenamic acid (Ponstan), may cause interstitial nephritis, glomerulitis, papillary necrosis and the nephrotic syndrome. NSAIDs inhibit the synthesis of renal prostaglandin which plays a supportive role in the maintenance of renal perfusion in patients whose renal blood flow and blood volume are decreased. In these patients, administration of an NSAID may precipitate overt renal decompensation, which is typically followed by recovery to pretreatment state upon discontinuation of NSAID therapy. Patients at greatest risk of such a reaction are those with congestive heart failure, liver cirrhosis, nephrotic syndrome, overt renal disease and the elderly. Such patients should be carefully monitored while receiving NSAID therapy.
Discontinuation of NSAID therapy is typically followed by recovery to the pre-treatment state. Since mefenamic acid metabolites are eliminated primarily by the kidneys, Ponstan should not be administered to patients with significantly impaired renal function.
Hematologic Effects: Ponstan can inhibit platelet aggregation and may prolong prothrombin time in patients on warfarin therapy (see Interactions).
Hepatic Effects: Borderline elevations of =1 liver function tests may occur in some patients receiving mefenamic acid (Ponstan) therapy. These elevations may progress, may remain essentially unchanged or may be transient with continued therapy. A patient with symptoms and/or signs suggesting liver dysfunction, or in whom an abnormal liver test has occurred, should be evaluated for evidence of the development of more severe hepatic reaction while on therapy with mefenamic acid (Ponstan). If abnormal liver tests persist or worsen, if clinical signs and symptoms consistent with liver disease develop, or if systemic manifestations occur, mefenamic acid (Ponstan) should be discontinued.
Use with Oral Anticoagulants: The concomitant use of NSAIDs, including mefenamic acid, with oral anticoagulants increases the risk of GI and non-GI bleeding and should be given with caution. Oral anticoagulants include warfarin/coumarin-type and novel oral anticoagulants (eg, apixaban, dabigatran, rivaroxaban). Anticoagulation/INR should be monitored in patients taking a warfarin/coumarin-type anticoagulant (see Interactions).
Effects on the Ability to Drive or Operate Machinery: The effect of mefenamic acid (Ponstan) on the ability to drive or use machinery has not been systematically evaluated.
Impairment of Fertility: Based on the mechanism of action, the use of NSAIDs may delay or prevent rupture of ovarian follicles, which has been associated with reversible infertility in some women. In women who have difficulties conceiving or who are undergoing investigation of infertility, withdrawal of NSAIDs, including mefenamic acid (Ponstan) should be considered.












Reviews
There are no reviews yet.