Description
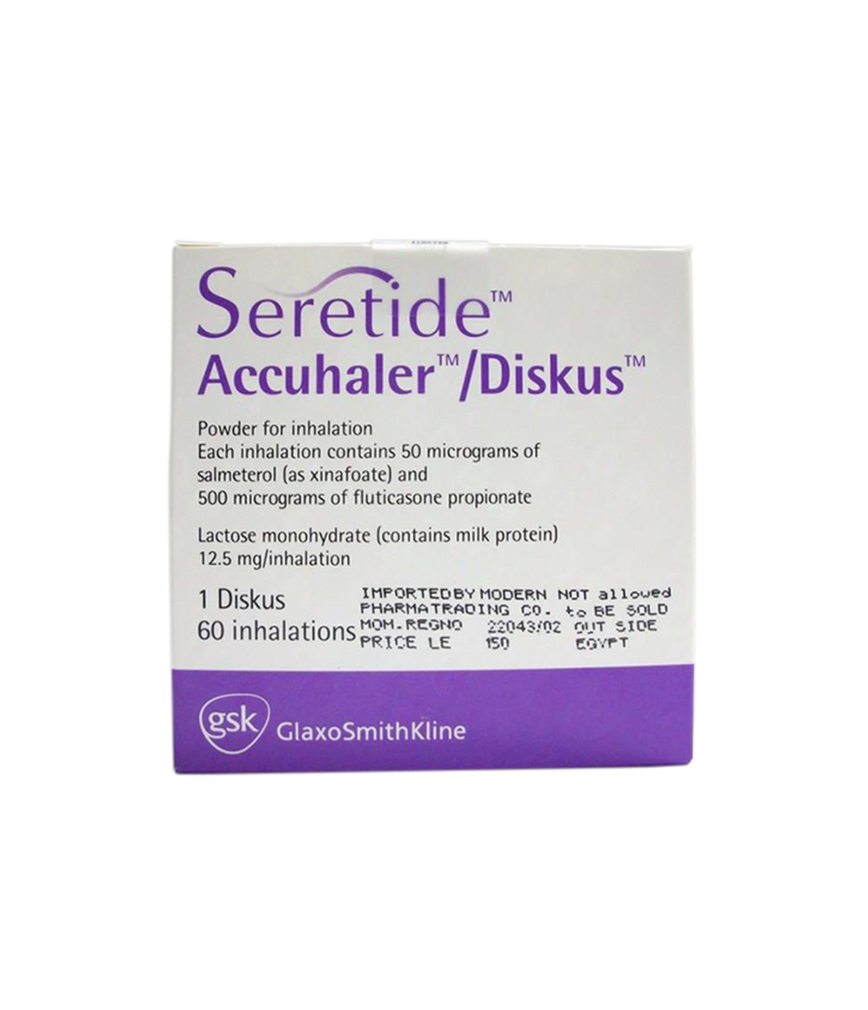
Buy Seretide Diskus 50/500 60D-PSP at Rose Pharmacy Online Drugstore
Indications / Uses :
Asthma [Reversible Obstructive Airway Disease (ROAD)]: Regular treatment of asthma may include: Patients on effective maintenance doses of long-acting ?-agonists and inhaled corticosteroids; who are symptomatic on current inhaled corticosteroid therapy; on regular bronchodilator therapy who require inhaled corticosteroids.
Chronic Obstructive Pulmonary Disease (COPD): Regular treatment of COPD including chronic bronchitis and emphysema.
Administration :
Contraindications : History of hypersensitivity to salmeterol, fluticasone propionate or any of the ingredients of Seretide.
Special Precautions :
Seretide is not for relief of acute symptoms for which a fast- and short-acting bronchodilator (eg, salbutamol) is required. Patients should be advised to have their relief medication available at all times.
Increasing use of short-acting bronchodilators to relieve symptoms indicates deterioration of control and patients should be reviewed by a physician.
Sudden and progressive deterioration in control of asthma is potentially life-threatening and the patient should be reviewed by a physician. Consideration should be given to increasing corticosteroid therapy. Also, where the current dosage of Seretide has failed to give adequate control of asthma, the patient should be reviewed by a physician.
Treatment with Seretide should not be stopped abruptly in patients with asthma due to the risk of exacerbation, therapy should be titrated down under physician supervision. For patients with COPD, cessation of therapy may be associated with symptomatic decompensation and should be supervised by a physician.
There was an increased reporting of pneumonia in studies of patients with COPD receiving Seretide (see Adverse Reactions). Physicians should remain vigilant for the possible development of pneumonia in patients with COPD as the clinical features of pneumonia and exacerbation frequently overlap.
As with all inhaled medication containing corticosteroids, Seretide should be administered with caution in patients with active or quiescent pulmonary tuberculosis.
Seretide Diskus 50/500 60D-PSP should be administered with caution in patients with thyrotoxicosis.
Cardiovascular effects eg, increases in systolic blood pressure and heart rate, may occasionally be seen with all sympathomimetic drugs, especially at higher than therapeutic doses. For this reason, Seretide should be used with caution in patients with preexisting cardiovascular disease.
A transient decrease in serum potassium may occur with all sympathomimetic drugs at higher therapeutic doses. Therefore, Seretide should be used with caution in patients predisposed to low levels of serum potassium.
Systemic effects may occur with any inhaled corticosteroid, particularly at high doses prescribed for long periods; these effects are much less likely to occur than with oral corticosteroids (see Overdosage). Possible systemic effects include Cushing’s syndrome, Cushingoid features, adrenal suppression, growth retardation in children and adolescents, decrease in bone mineral density, cataract and glaucoma. It is important therefore for asthma patients, that the dose of inhaled corticosteroid is titrated to the lowest dose at which effective control is maintained.
The possibility of impaired adrenal response should always be borne in mind in emergency and elective situations likely to produce stress and appropriate corticosteroid treatment considered (see Overdosage).
It is recommended that the height of children receiving prolonged treatment with inhaled corticosteroid is regularly monitored.
Because of the possibility of impaired adrenal response, patients transferring from oral steroid therapy to inhaled fluticasone propionate therapy should be treated with special care and adrenocortical function regularly monitored.
Following introduction of inhaled fluticasone propionate, withdrawal of systemic therapy should be gradual and patients encouraged to carry a steroid warning card indicating the possible need for additional therapy in times of stress.
There have been very rare reports of increases in blood glucose levels (see Adverse Reactions) and this should be considered when prescribing to patients with a history of diabetes mellitus.
During post-marketing use, there have been reports of clinically significant drug interactions in patients receiving fluticasone propionate and ritonavir, resulting in systemic corticosteroid effects including Cushing’s syndrome and adrenal suppression. Therefore, concomitant use of fluticasone propionate and ritonavir should be avoided, unless the potential benefit to the patient outweighs the risk of systemic corticosteroid adverse effects (see Interactions).
Data from a large US study (SMART) comparing the safety of Serevent, (a component of Seretide) or placebo added to usual therapy showed a significant increase in asthma-related deaths in patients receiving Serevent. Data from this study suggested that African-American patients may be at greater risk of serious respiratory-related events or deaths when using Serevent compared to placebo. It is not known if this was due to pharmacogenetic or other factors. The SMART study was not designed to determine whether concurrent use of inhaled corticosteroids modifies the risk of asthma-related death (see Pharmacology: Clinical Studies under Actions).
It was observed in a drug interaction study that concomitant use of systemic ketoconazole increases exposure to Serevent. This may lead to prolongation in the QTc interval. Caution should be exercised when strong CYP3A4 inhibitors (eg, ketoconazole) are co-administered with Serevent (see Pharmacology: Pharmacokinetics under Actions and Interactions).
As with other inhalation therapy, paradoxical bronchospasm may occur with an immediate increase in wheezing after dosing. This should be treated immediately with a fast- and short-acting inhaled bronchodilator. Salmeterol-fluticasone propionate accuhaler/diskus or evohaler should be discontinued immediately, the patient assessed and alternative therapy instituted if necessary (see Adverse Reactions).
The pharmacological side effects of ?2-agonist treatment eg, tremor, subjective palpitations and headache have been reported, but tend to be transient and to reduce with regular therapy (see Adverse Reactions).
Effects on the Ability to Drive or Operate Machinery: There have been no specific studies of the effect of Seretide on the previously mentioned activities, but the pharmacology of both drugs does not indicate any effect.
Use in Children: There are no data available for use in Seretide in children <4 years.
Use in Pregnancy & Lactation: Administration of drugs during pregnancy and lactation should only be considered if the expected benefit to the mother is greater than any possible risk to the fetus or child.
There is insufficient experience of the use of salmeterol xinafoate and fluticasone propionate in human pregnancy and lactation.
Reproductive toxicity studies in animals, either with single drug or in combination, revealed the fetal effects expected at excessive systemic exposure levels of a potent ?2-adrenoreceptor agonist and glucocorticosteroid.
Extensive clinical experience with drugs in these classes has revealed no evidence that the effects are relevant at therapeutic doses. Neither salmeterol xinafoate or fluticasone propionate have shown any potential for genetic toxicity.
Salmeterol and fluticasone propionate concentrations in plasma after inhaled therapeutic doses are very low and therefore, concentrations in human breast milk are likely to be correspondingly low. This is supported by studies in lactating animals, in which low drug concentrations were measured in milk. There are no data available for human breast milk.












Reviews
There are no reviews yet.